IN A NUTSHELL
The duality of JAK-inhibition: Dampening autoimmunity and promoting the regeneration of bone
Affecting up to 1% of the global population, rheumatoid arthritis is the most common inflammatory joint disease. In rheumatoid arthritis, as a chronic autoimmune disease, the body’s own immune system falsely identifies the body’s own tissues as “foreign”. When this happens, immune cells infiltrate the joint cavity and synovium, where they induce painful inflammation and swelling. When left untreated, rheumatoid arthritis irreversibly destroys cartilage and bone and will ultimately result in immobility. Unfortunately there is still no cure for rheumatoid arthritis, but treatment options are constantly being improved. Among these, a new class of drug has emerged, the Janus kinase “JAK” inhibitors. JAKs belong to a ubiquitous signaling pathway and facilitate the intracellular response to extracellular mediators. Some of these mediators are involved in the pathogenesis of rheumatoid arthritis, which is why JAK inhibitors are approved as a treatment for rheumatoid arthritis.
An unaddressed issue in rheumatoid arthritis is irreparable tissue deterioration because current therapies are unable to restore bone tissue once it is eroded. However, JAK inhibitors might shift this paradigm. Despite their approval for the treatment of rheumatoid arthritis, their impact on bone metabolism has not yet been systematically investigated. Therefore, we explored the influence of JAK inhibitors on bone homeostasis and unraveled their clinical significance as osteoanabolic agents, which increase and maintain bone mass during, but also in the absence of, inflammation.
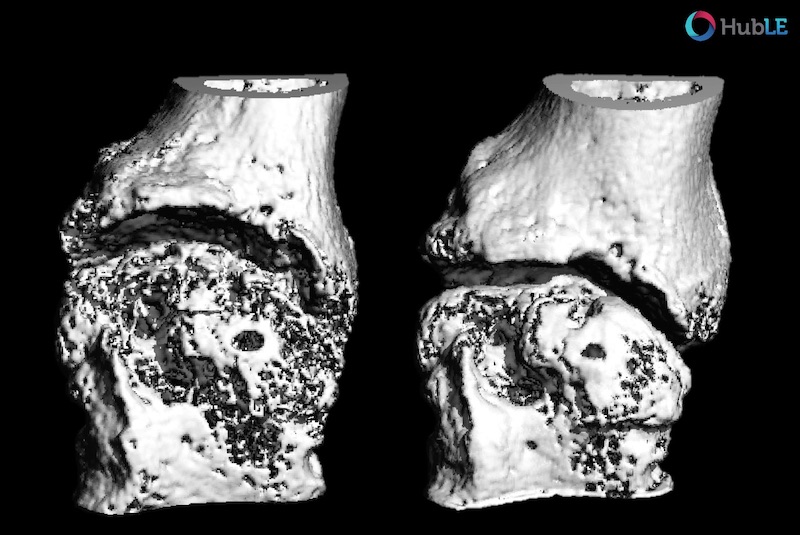
To evaluate if (non)inflammatory pathological challenges affect the impact of JAK inhibition on bone, we tested JAK inhibition in different preclinical mouse models: (1) Healthy steady-state conditions, (2) an arthritis-model of inflammation-mediated bone loss and (3) an osteoporosis-model with ovariectomy-induced bone-loss. JAK inhibition ameliorated inflammation-mediated disease manifestation, and also increased bone mass consistently, regardless of disease background. In vitro analysis further demonstrated that JAK inhibition directly enhances the ability of bone-forming osteoblasts to produce bone matrix, and whole transcriptome-analysis revealed an increased Wnt signature in JAK inhibition-treated osteoblasts – the Wnt signaling pathway being a central promoter of bone formation. Lastly, two rheumatoid arthritis patients who received tofacitinib (a JAK inhibitor) were examined for bone morphology in their metacarpophalangeal joints (the joints at the base of the fingers) by high-resolution peripheral quantitative computed tomography. Both patients clearly demonstrated signs of bone formation after tofacitinib-therapy. In summary, our data revealed that JAK inhibition increases bone mass in steady-state conditions and during pathological challenges.
Thus, can JAK-inhibition serve as a universal cure for osteoporotic diseases? Unfortunately, it is not that simple. JAK inhibition definitely has promising potential as an osteoanabolic treatment. However, the role of JAK as a universal signaling molecule cannot be neglected. Diseases may be treated more efficiently by targeting multiple cytokines simultaneously but adverse reactions are more likely to occur. Therefore, the patient’s medical background and the potential benefit of JAK inhibition must be carefully evaluated to assess the risk-benefit ratio for treating bone-loss diseases.
The capability of JAK-inhibitors to directly impact bone metabolism will expand their well-established role as anti-inflammatory agents. Bone loss represents a feature of rheumatoid arthritis, and osteoporosis is a common comorbidity in rheumatoid arthritis patients. With JAK inhibitors already being an approved treatment for rheumatoid arthritis, these findings might have immediate benefits for patients with rheumatoid arthritis. In addition, while the preliminary human data makes us confident for JAK inhibition to prove its bone-forming benefit, clinical studies are indispensable to evaluate its scope. Therefore, we are looking forward to future clinical studies, as are being performed by Dr. Arnd Kleyer at the medical department at the university hospital in Erlangen.
These findings are described in the article entitled “JAK inhibition increases bone mass in steady-state conditions and ameliorates pathological bone loss by stimulating osteoblast function” published in Science Translational Medicine. This work was conducted by Susanne Adam, Nils Simon, Ulrike Steffen, Fabian T. Andes, Carina Scholtysek, Dorothea I. H. Müller, Daniela Weidner, Darja Andreev, Arnd Kleyer, Stephan Culemann, Madelaine Hahn, Georg Schett, Gerhard Krönke, Silke Frey and Axel J. Hueber from the Friedrich-Alexander-University Erlangen-Nürnberg (FAU) and the Universitätsklinikum Erlangen (Germany). It was supported by the Deutsche Forschungsgemeinschaft (FOR 2886 PANDORA, DFG-CRC1181, SCHE (1583/14-1), HA 8163/1-1), research grants from Eli Lilly and Pfizer, the Bundesministerium für Bildung und Forschung (METARTHROS), the European Union (ERC StG 640087–SOS, ERC 4D NanoScope) and the EU/EFPIA Innovative Medicines Initiative 2 Joint Undertaking RTCure grant 777357, and the Interdisciplinary Center for Clinical Research at the Universitätsklinikum Erlangen of the FAU. The authors thank the staff, contributors and participants involved in this project.

Susanne Adam, PhD
Universitätsklinikum Erlangen, Germany.