THESIS REPORT
Role of IKKα in prostate cancer bone metastases
Abdullah Aljeffrey
July 2019
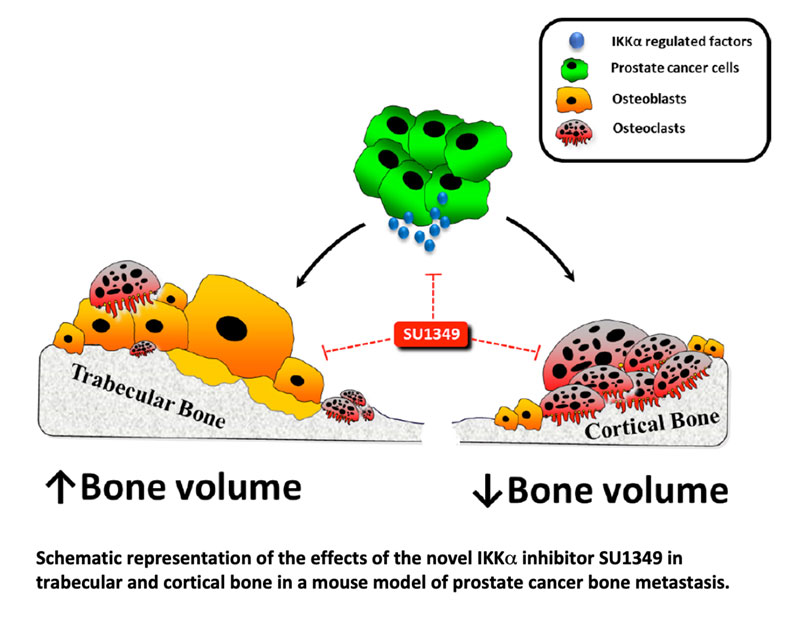
IκB kinase subunit alpha (IKKa), a key component of the NFκB pathway, is implicated in prostate cancer progression and bone remodelling, but its role in the regulation of prostate cancer associated bone disease remains unknown. Here, we tested the effects of IKKamanipulation on prostate cancer cell behaviour in vitro and on osteolysis in a xenograft model of human prostate cancer bone metastasis. The pharmacological inhibition of IKKausing the novel and highly selective IKKainhibitor SU1349 and stable knockdown of IKKain PC3 cells significantly reduced cell viability, migration and invasion, whereas IKKa overexpression was stimulatory. In prostate cancer cell – osteoclast co-cultures, IKKaoverexpression in PC3 enhanced RANKL-induced osteoclastogenesis, whereas SU1349 treatment and IKKa knockdown reduced these effects. Exposure of osteoblasts to SU1349 enhanced alkaline phosphatase activity and bone nodule formation. Protein microarray analysis of tumour-derived factors in PC3 conditioned medium showed that these effects were associated with significant inhibition of various NFκB-mediated pro-inflammatory factors and reduction in PC3 conditioned medium induced NFκB activation in osteoclast and osteoblast precursors. In osteoclast and osteoblast precursors SU1349 lead to inhibition of both canonical and non-canonical NFκB signalling. Interestingly, NFκB inhibition in osteoblasts was accompanied by an increase in GSK3β phosphorylation indicative of activation of the Wnt/β-catenin pathway, whereas the opposite occurred in osteoclast precursors. In vivo, administration of SU1349 (20mg/kg/3times-weekly) in immuno-deficient BALB/c mice after intra-cardiac injection with human PC3 cells enhanced trabecular bone volume and this was associated with reduced osteoclast and increased osteoblast numbers. Paradoxically, SU1349 treatment reduced cortical bone volume, and this was associated with increased osteoclast and reduced osteoblast numbers. Thus, the novel IKKa inhibitor SU1349 is of potential therapeutic value in protecting against prostate cancer-induced osteolysis. However, exacerbation of bone loss in the cortical compartment may limit its usefulness as a bone sparing agent.
Full text of Dr A. Aljeffery’s thesis will be available in March 2019.

Abdullah Aljeffrey
University of Sheffield, UK

