HUBLE METHODS
Culture of the IDG-SW3 osteocyte cell line
Introduction
The IDG-SW3 osteoblast-to-late-osteocyte cell line is derived from a temperature-sensitive DMP1-GFP transgenic mouse. IDG-SW3s cultured with interferon g (IFNγ) at 33oC will proliferate, whilst culture in mineralising conditions without IFNγ at 37oC enhances differentiation. This HubLE Method describes the protocol for culturing this cell line [1].
Materials
- Rat-tail type 1 collagen
- Recombinant mouse interferon g (IFNγ)
- Phosphate buffered saline (PBS)
- Proliferation medium Alpha Modified Essential Medium (ProlifαMEM) [Tip No. 1]
- Osteocyte-differentiation αMEM (OcyMEM) [Tip No. 2]
- Trypsin-EDTA (0.25%)
- Glutaraldehyde (2.5%)
- Alizarin Red (1%)
Methods [Update]
- Coat all required tissue culture plastics for 1 hour at room temperature with 0.15mg/ml of rat-tail type 1 collagen in 0.02M acetic acid.
- Remove the collagen solution and either wash with PBS for immediate use, or air dry plates prior to storage [Tip No. 3].
- Thaw a vial of IDG-SW3 cells into 5ml ProlifαMEM and spin at 1,500 rpm for 5 minutes.
- Remove the supernatant, re-suspend the pellet in ProlifαMEM and seed into a collagen-coated 75cm2 flask containing ProlifαMEM. Incubate at 33oC with 5% CO2.
- Once ≥80% confluent (2-3 days post-seeding), remove medium, wash with PBS and incubate with 0.25% Trypsin for 5-10 minutes to detach cells.
- Spin at 1,500g for 5 minutes and re-suspend in ProlifαMEM (1ml per flask).
- Seed cells in collagen-coated tissue culture trays or flasks (for further expansion and use of cells at next passage) in ProlifαMEM [Tip No. 4].
- Incubate at 33oC with 5% CO2 until confluent.
- At this stage, remove the ProlifαMEM medium, carefully wash the cell monolayers with PBS and add OcyMEM. Incubate at 37oC with 5% CO2.
- Culture for up to 30-35 days with half medium changes of OcyMEM every 2-3 days. Mineralisation is usually evident from ~10-14 days.
- SFix with 2.5% glutaraldehyde for 5 minutes before staining with 1% alizarin red to visualise mineralised nodules [Tip No. 5].
Tips [Update]
ProlifαMEM: Add 10% heat-inactivated foetal calf serum (FCS), AB/AM (100U/ml penicillin, 100mg/ml streptomycin, 0.25mg/ml amphotericin) and L-glutamine (200mM). Aliquot the stock media and add IFNγ (2500U/ µl). Incubate the medium at 33oC prior to use and limit exposure to heat due to IFNγ degradation.
OcyMEM: Add 10% heat-inactivated foetal calf serum (FCS), AB/AM (100U/ml penicillin, 100mg/ml streptomycin, 0.25mg/ml amphotericin) and L-glutamine (200mM). Add 50µg/ml ascorbate and 2-4mM β-glycerophosphate (the original paper uses 4mM β-GP [1], however, IDG-SW3 cells differentiate and mineralise sufficiently in 2mM). Always make fresh on the day of use.
Collagen-coated tissue culture plates/ flasks: Ensure all plates are coated under sterile conditions in a tissue culture hood. Use a cold pipette (stored in the freezer until use) to stop the collagen sticking to the plastic. The 0.15mg/ml collagen solution can be re-used 5-6 times; coating for ~1 hour each time. Coated plastics wrapped in parafilm can be stored at 4oC for up to 6 months until use.
Seeding density: IDG-SW3 cells will mineralise in 12 and 6-well plates but due to the long culture duration some monolayer peeling should be expected. Woo et al. [1] recommend seeding IDG-SW3 cells at 4×104 cells/cm2, although the lower densities of 104 (12-well) and 105 (6-well) will also support osteocyte proliferation, mineralisation and differentiation. To expand IDG-SW3 cells for the subsequent passage seed at 5×105 cells/ 75cm2 flask.
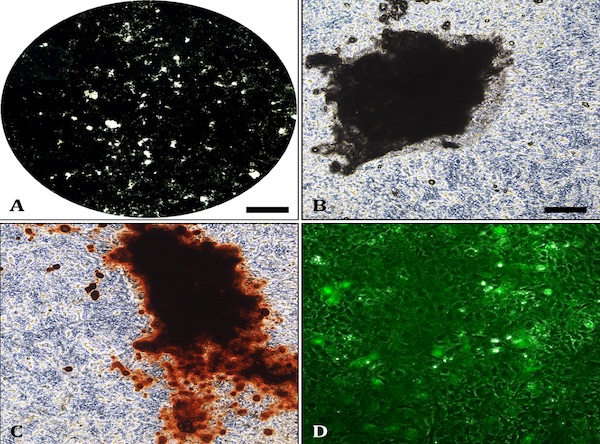
Alizarin Red staining: Mineralised bone nodules can be stained with alizarin red (Fig.1C). It is also possible to obtain good quality images on unstained cell layers (Fig 1A-1B). DMP1-GFP expression can be visually monitored throughout the differentiation process (Fig.1D). Evaluation of E11, DMP1 and sclerostin gene/ protein expression is also advisable to confirm osteocyte differentiation.
References [Update]
Woo SM, Rosser J, Dusevich V, Kalajzic I, Bonewald LF (2011). Cell line IDG-SW3 replicates osteoblast-to-late osteocyte differentiation in vitro and accelerates bone formation in vivo. J Bone Mineral Res 26:2634-2646.

Bethan K Davies, MBio
Department of Comparative Biomedical Sciences,
Royal Veterinary College, London. NW1 0TU, UK.
Email: bkdavies@rvc.ac.uk

Isabel R Orriss, BSc, PhD
Department of Comparative Biomedical Sciences,
Royal Veterinary College, London, NW1 0TU, UK
Email: iorriss@rvc.ac.uk